AI Medical Jenga: From Abstraction to Physiology
- Apr 6
- 4 min read

The temptation in computational biology is to believe that a well-behaved model is a truthful one. It is not. A model is only as honest as the physiological commitments it makes—and more importantly, the ones it avoids.
If you have been following our recent work in the AI Medical System Jenga Optimisation notebook, you know we began with a fascinating control problem. We modelled an agent balancing a metaphorical game of Jenga: managing "ATP" as a resource, treating "Stability" as survival, and intervening with arbitrary "PULL" actions on a structure that degrades over time.
That is a valid mathematical abstraction. But biology is not an abstraction—it is a constraint. The task now is not to rename variables, but to bind them to measurable, falsifiable physiology. Here is how we transition our toy control system into Physiologically Based Pharmacokinetic (PBPK) reality.
1. The Translation Problem: What Are We Actually Modelling?
At its core, our original system described a dynamic balance between energy production, structural integrity, and intervention cost. In pharmacokinetics, this translates directly into a system's ability to process and eliminate a drug (Clearance), access to compartments (Bioavailability), and tolerance to perturbation (Physiological reserve).
The critical shift is this: we are no longer modelling "a system that survives". We are modelling a system that clears drug under constraint.
2. Recasting "ATP" (Energy is Not a Number)
In our toy model, the variable "ATP" is a bounded scalar with arbitrary increments. But in physiology, energy is not a number—it is a network state.
Instead of an abstract health bar, we must choose a measurable proxy. The most actionable choice for PBPK modeling is mapping ATP to intrinsic clearance (CLint).
Clearance is not just flow—it is capacity.
Capacity is energy-dependent.
Energy failure manifests as reduced metabolic turnover.
3. Clearance as the Central Axis
At the heart of this translation lies one equation:
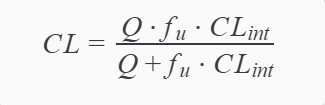
Where

is hepatic blood flow,

is fraction unbound, and

is intrinsic clearance. This is not just a formula—it is a statement of biological dependency. If

drops, clearance becomes capacity-limited. If

drops, clearance becomes flow-limited. If both degrade, you face system collapse. Our "ATP" variable now has a place: it modulates

, and therefore the organism's ability to process a drug.
4. Redefining "Stability" and "Jenga Pulls"
"Stability" in the original model meant structural integrity. In physiology, we must define what failure actually means. The most relevant metric for PBPK integration is mapping stability to perfusion

and organ function, measured by clinical vitals like Mean Arterial Pressure (MAP).
Similarly, our original reinforcement-learning actions (WAIT, PULL LOW, PULL MID, PULL HIGH) must be translated into dosing decisions:
WAIT → No dosing / Washout
PULL LOW → Low dose
PULL MID → Therapeutic dose
PULL HIGH → Supratherapeutic / Toxic dose
Our AI agent is no longer playing a game—it is selecting dosing strategies under physiological constraint.
5. The Feedback Loop: When the Drug Fights Back
This is where our model becomes genuinely interesting. We must move away from the assumption that the "drug is processed by the system" and embrace the reality that the "drug changes the system that processes it".
Drugs can alter clearance through several mechanisms:
Enzyme inhibition: Reduces

(e.g., ketoconazole inhibiting CYP3A4)
Hepatotoxicity: Reduces functional hepatocyte mass, decreasing

and possibly

Mitochondrial toxicity: Reduces ATP production, indirectly reducing metabolic capacity
By defining metabolic state as a function of drug exposure, we create a vicious loop: drug concentration causes metabolic stress, which reduces clearance, which leads to even higher drug concentration. This is the beginning of nonlinear pharmacokinetics.
6. Optimising for the Clinic, Not the Arcade
Finally, our arbitrary reward system must be replaced with measurable clinical outcomes. A successful model balances therapeutic exposure (AUC, Time above threshold) against toxicity risk (Cmax) and system degradation (Lactate, MAP). This is no longer a simple reward function—it is a clinical optimisation problem.
Final Reflection
If this translation feels messy, that is correct. Biology is not clean. What we have done is subtle but profound: we replaced a symbolic resource with a mechanistic determinant of clearance, and transformed a reinforcement-learning toy into a constrained pharmacological system with feedback.
A model becomes scientific not when it is complex, but when it is accountable—to measurement, to mechanism, and to failure. In our original system, collapse was defined by rules. In physiology, collapse is discovered through consequence. That is the new standard the AI must meet.
Related Models:



Comments